|
12/31/2023 0 Comments Keynote 042 
However, nearly half of patients with PD-L1 ≥1% and approximately 75% of patients with PD-L1 ≥20% had expression ≥50% on TCs. The basis for the FDA approval was that pembrolizumab demonstrated a significantly improved OS in KEYNOTE-042 when compared to platinum-based doublets for patients with PD-L1 ≥50%, ≥20% and ≥1% on TCs ( 15). However, a detailed analysis of KEYNOTE-042 raises the possibility that pembrolizumab monotherapy may not be the best option for many patients with advanced/metastatic NSCLC who lack EGFR activating mutations/ ALK fusions and have PD-L1 of 1–49% on TCs. Subsequently, on April 11 th 2019 the Food and Drug Administration (FDA) approved pembrolizumab monotherapy for NSCLC patients lacking EGFR activating mutations/ ALK fusions expressing PD-L1 ≥1% on TCs who are metastatic or have stage III disease not felt appropriate for surgery/chemoradiation ( 15). The trial headline was that pembrolizumab monotherapy might be an additional effective option for this patient population ( 14). KEYNOTE-042 compared pembrolizumab to histology dependent platinum-based doublets as initial treatment in locally advanced or metastatic patients with PD-L1 ≥1% on TCs and lacking EGFR activating mutations/ ALK fusions. 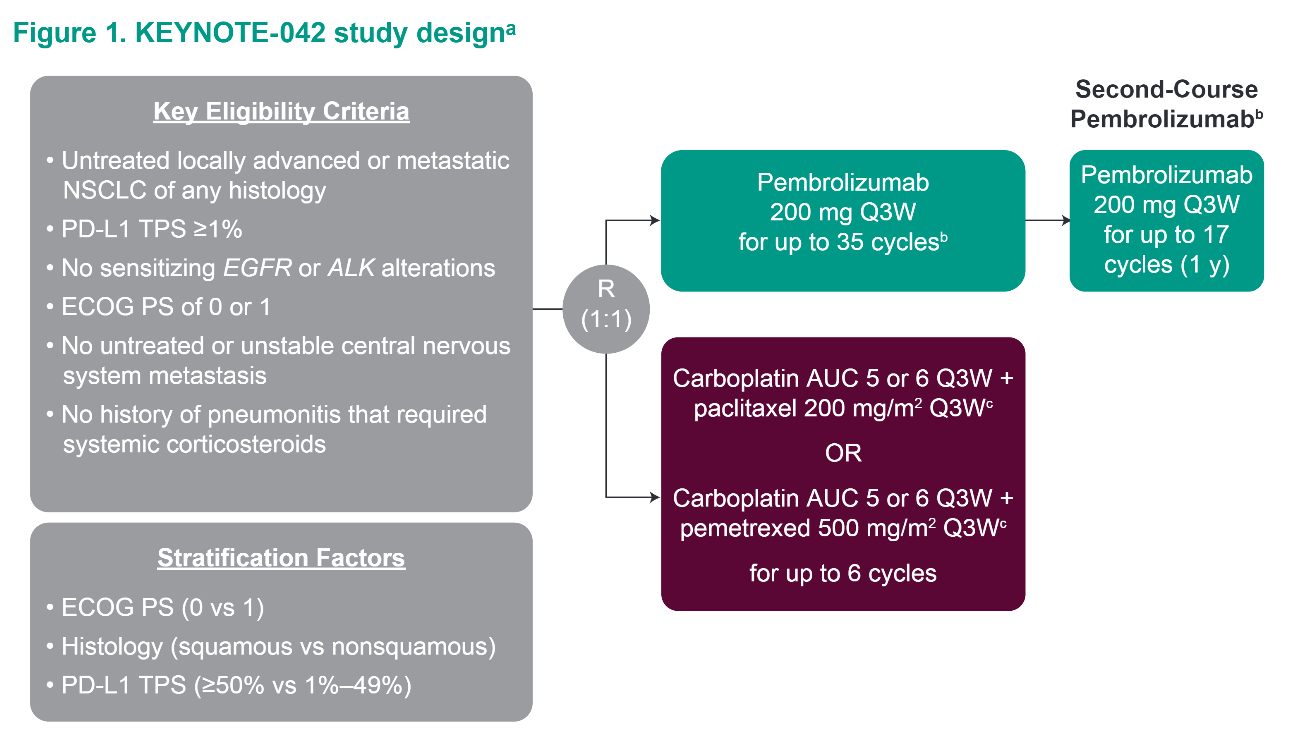
Until recently, the recommended first-line therapy by most guideline panels and regulatory agencies for metastatic NSCLC patients lacking an approved targeted therapy option has been pembrolizumab monotherapy or chemo-immunotherapy (ABCP or pembrolizumab plus histology dependent chemotherapy) if PD-L1 was ≥50% on TCs and chemo-immunotherapy if PD-L1 was <50% on TCs. CheckMate-026, which compared nivolumab to histology dependent platinum-based doublets in patients without EGFR activating mutations/ ALK fusions, failed to demonstrate improved OS for nivolumab in the overall study population with PD-L1 ≥5% on TCs or in the subgroup with PD-L1 ≥50% on TCs ( 13). Similarly, within KEYNOTE-407, pembrolizumab plus platinum/taxane demonstrated improved ORR (57.9% vs. 48%), PFS (HR 0.62, P<0.001) and OS (HR 0.78, P=0.02) for non-squamous NSCLC patients regardless of PD-L1 staining when compared to carboplatin plus paclitaxel plus bevacizumab ( 12). In IMpower150, the regimen of ABCP also demonstrated improved ORR (63.5% vs. 18.9%, P<0.001), PFS and OS (HR 0.49, P<0.001) than platinum/pemetrexed across non-squamous NSCLC patients with any level PD-L1 staining (including PD-L1 negative patients) ( 10). In KEYNOTE-189, pembrolizumab plus platinum/pemetrexed had better ORR (47.6% vs. For patients lacking EGFR activating mutations/ ALK fusions these chemo-immunotherapy combinations improved efficacy outcomes compared to platinum-based chemotherapy ( 10- 12). Following this, both atezolizumab plus bevacizumab plus carboplatin plus paclitaxel (ABCP) and pembrolizumab plus histology dependent chemotherapy were compared to platinum-based chemotherapy in randomized phase III trials. Subsequently, KEYNOTE-024 demonstrated that for advanced/metastatic NSCLC patients with PD-L1 ≥50% on TCs by the 22-C3 immunohistochemistry assay and lacking EGFR activating mutations/ ALK fusions that pembrolizumab had a significantly greater objective response rate (ORR), progression-free survival (PFS) and OS when compared to platinum-based doublets as first-line therapy ( 8, 9).īecause of KEYNOTE-024, multiple regulatory agencies and guideline panels approved pembrolizumab monotherapy for patients with newly diagnosed advanced/metastatic NSCLC, PD-L1 ≥50% on TCs and lacking EGFR activating mutations/ ALK fusions. In each of these second-line trials patients with epidermal growth factor receptor (EGFR) activating mutations did not benefit from PD-1 axis inhibitors when compared to docetaxel, with data on any included ALK rearranged patients not detailed ( 4- 7). 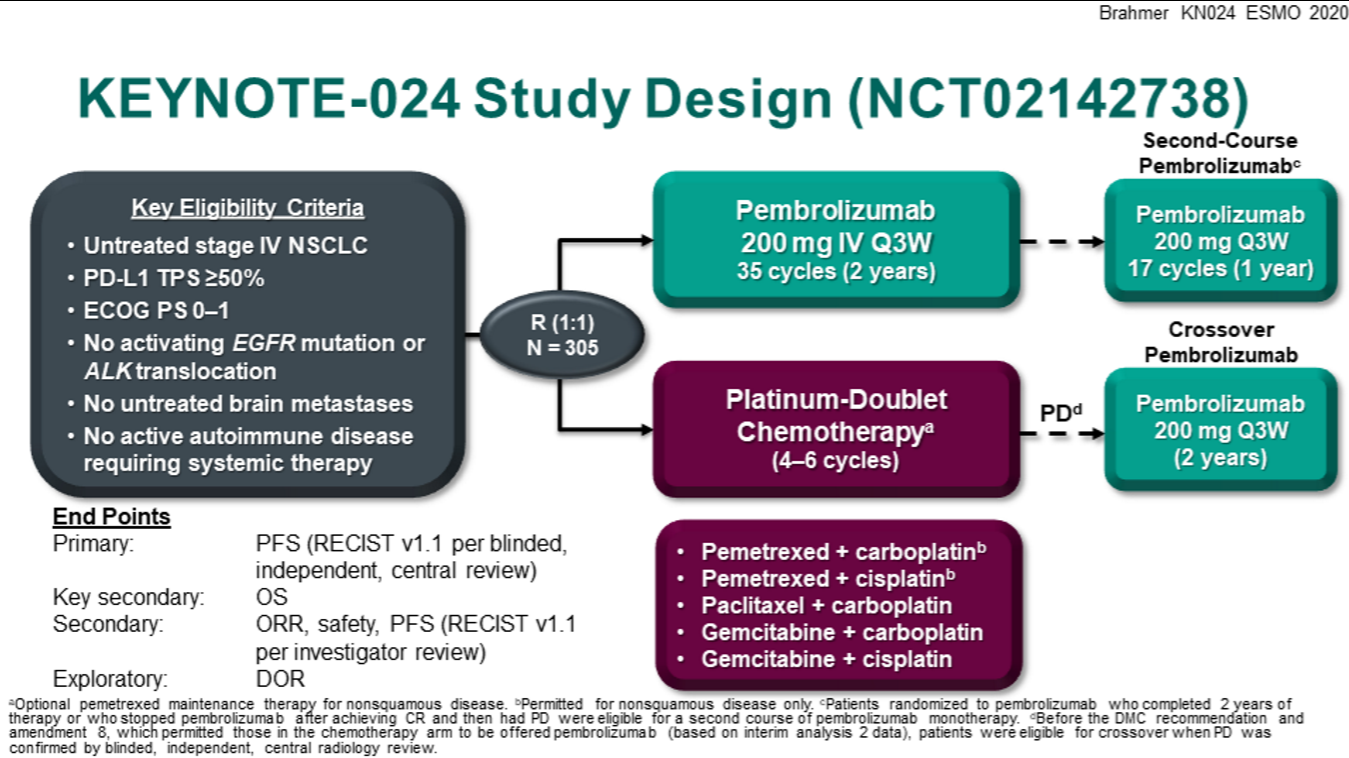
Within KEYNOTE-010, patients with PD-L1 ≥50% on tumor cells (TCs) administered pembrolizumab had a greater magnitude of benefit compared to docetaxel than patients with PD-L1 of 1–49% ( 6). In each of these trials, there was an OS benefit when comparing the respective PD-1 or PD-L1 inhibitor to docetaxel ( 4- 7). Subsequently, randomized phase III trials comparing atezolizumab, nivolumab or pembrolizumab to docetaxel as second-line therapy were conducted in NSCLC. Prior to the advent of inhibitory antibodies targeting programmed death 1 (PD-1) or programmed death ligand 1 (PD-L1), the median overall survival (OS) for advanced/metastatic non-small cell lung cancer (NSCLC) patients receiving first-line platinum doublet chemotherapy was approximately 8–12 months and 5-year survival rates were estimated at 2% ( 1- 3). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
